Tricuspid valve surgery is a common heart valve procedure in the United States, but it is one with poor long-term outcomes. In fact, studies show that up to 45 percent of those who receive tricuspid valve surgery suffer a recurrence of problems, in some cases as early seven years after surgery. In one-third of the cases, a second operation is required.
But little is understood about the process by which repaired tricuspid valves change following surgery. Rouzbeh Amini, Ph.D., is trying to shed light those changes.
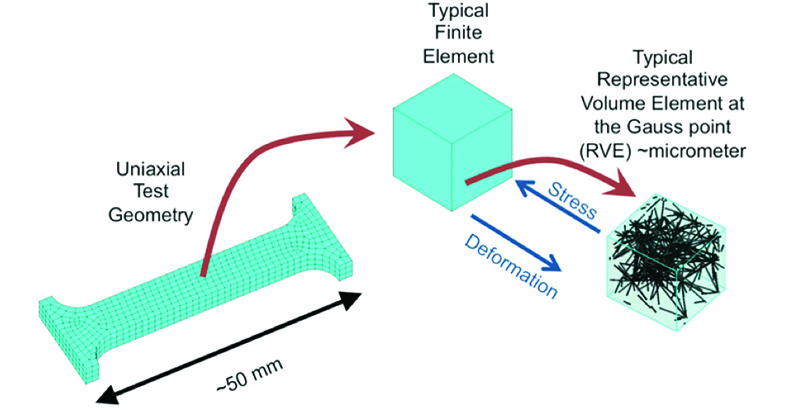
The University of Akron’s assistant professor in the Department of Biomedical Engineering is using the Ohio Supercomputer Center’s Oakley Cluster to develop a finite element model that can discover why tricuspid valves aren’t holding up long-term.
The tricuspid valve—which prevents backflow of blood into the right atrium of the heart—is the most understudied of the four valves in the heart, mostly because the left side of the heart is where most problems occur. Tricuspid valves often need repair surgeries when the valve doesn’t close completely, thus not guaranteeing one-way flow, or when the valve doesn’t open fully to allow enough blood flow.
After years of focusing on the left side, clinicians and surgeons started to notice tricuspid valve surgery wasn’t as successful as they had once hoped it would be.
Discovering why requires a detailed mathematical framework that can quantify how the fibers of the proteins elastin and collagen, which mainly make up the leaflets of the valve, react to the stretching and stresses needed for the valve to work.
“Imagine each one of these protein fibers as a single spring,” Amini said. “You stretch it, it resists. You can model a network of springs. The problem is it requires a great deal of computational resources.
“If you apply a certain amount of load to the valve, mechanical load, you want to know how the leaflets perform.”
Because Amini is dealing with a gargantuan amount of protein fibers and the structure in the tissue is complex, the computational aspect is intense. Amini and his team use codes composed in “C” to run finite element analysis through Oakley on the mathematical framework they’ve developed.
“Because of OSC, we don’t have to maintain the hardware and software, we can just focus on what we’re good at which is writing codes and translating biological problems into computational models,” Amini said.
###
Written by Ross Bishoff
Project Lead: Rouzbeh Amini, Ph.D., assistant professor biomedical engineering, University of Akron
Research Title: A deterministic multi-scale finite element model of tricuspid valve leaflets
Funding Source: American Heart Association
Website: http://amini.uakron.edu